Strategy
A skincare feasibility review clarifies ingredient logic, claims feasibility and development risks before formulation begins.

Nadia Zarrouk
Most skincare projects don’t go off track because a laboratory can’t formulate the product.
They go off track earlier when the concept is still vague and the development direction hasn’t been clearly defined.
A skincare feasibility review exists to solve that problem.
It transforms a product idea into a technically coherent direction before formulation begins, when decisions are still flexible and mistakes are inexpensive.

Where founders lose time without realising
Many founders move directly from product idea to contacting a laboratory.
On the surface, this seems efficient: describe the concept, request samples and begin development.
In practice, however, product development rarely unfolds so smoothly.
When the product direction hasn’t been fully clarified, early decisions begin to create unexpected technical consequences:
• a lightweight texture reduces structural stability
• an ingredient choice restricts packaging options
• a claim becomes difficult to support through testing
• preservation margins become tighter than expected
• timelines expand due to additional formulation rounds
None of these problems mean the product idea is flawed.
They simply indicate that key development decisions were never clarified at the start.
What a feasibility review actually examines
A feasibility review does not attempt to produce a finished formula.
Its purpose is to determine whether a product concept is technically coherent and strategically viable before formulation begins.
Rather than focusing on individual ingredients, the review evaluates the logic of the system the product will become, from formulation structure to claims feasibility and regulatory constraints.
The following areas are typically assessed.
Assessement area | Development implication |
Format logic | Determines the structural environment in which ingredients must function (serum, cream, oil, gel). |
Ingredient direction | Ensures active ingredients can coexist without instability, oxidation or preservation conflicts. |
Claims feasibility | Checks whether intended marketing claims can realistically be supported by formulation and testing. |
Regulatory considerations | Flags ingredient restrictions or labelling requirements across target markets such as the EU or UK. |
Development complexity | Identifies potential stability risks, testing requirements and formulation difficulty early in the process. |
The objective is simple:
to confirm that the concept can realistically become a product, before the laboratory begins formulation.
The Vitamin C Problem
Consider one of the most common skincare product ideas.
A founder wants to launch a Vitamin C serum positioned as:
brightening, antioxidant, premium texture, suitable for sensitive skin.
At first glance, the concept appears straightforward. Vitamin C is widely recognised, strongly associated with skin brightness, and frequently used in premium skincare products.
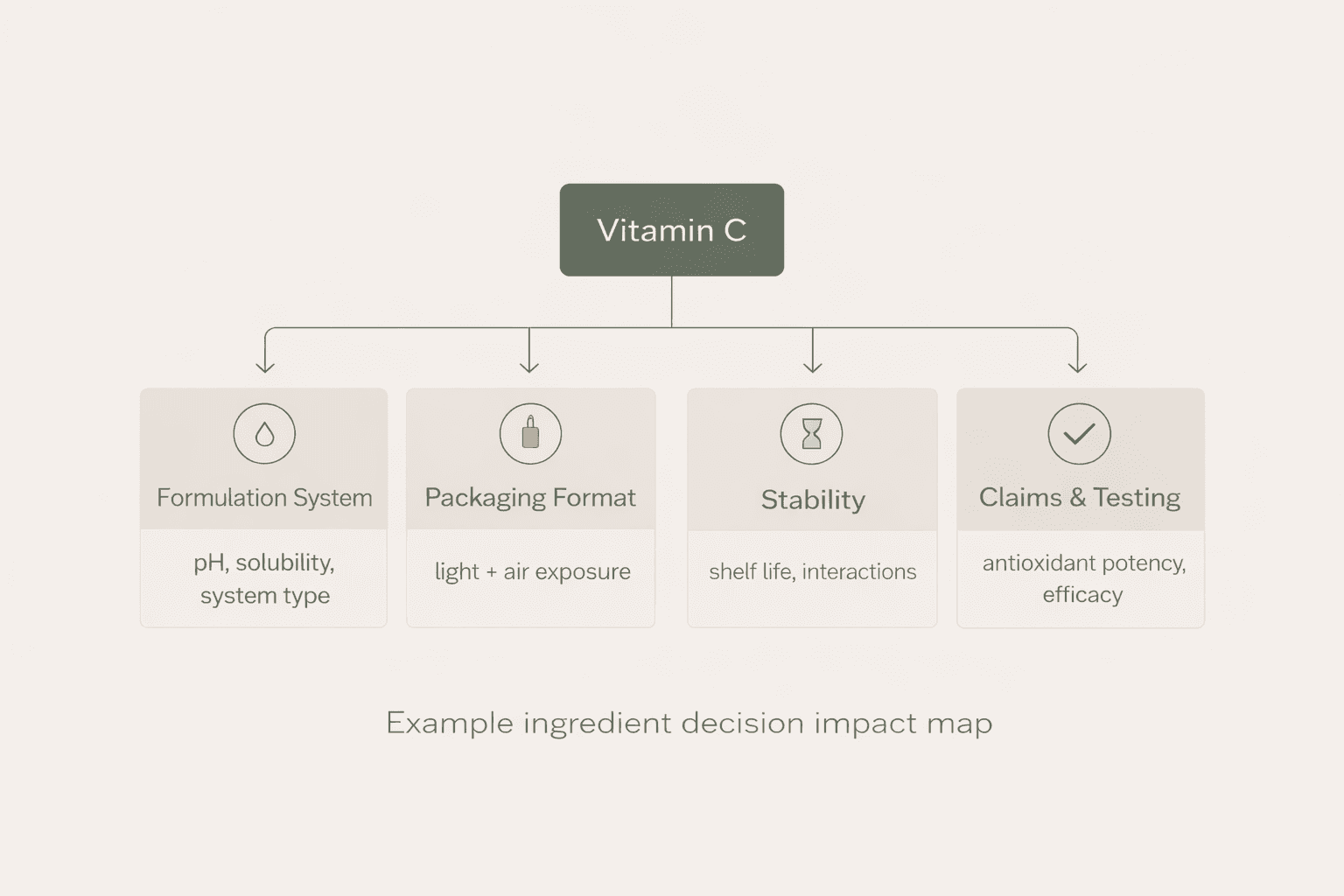
But once development begins, that single ingredient quickly influences a series of technical decisions.
The first challenge is stability.
Pure Vitamin C (ascorbic acid) is highly sensitive to oxygen, light and water exposure. If the formulation environment is not carefully controlled, oxidation can occur rapidly, leading to colour changes, reduced efficacy and a shorter shelf life.
Packaging therefore becomes part of the formulation strategy.
Airless pumps, opaque bottles or specialised packaging systems may be necessary to slow oxidation.
The second challenge is pH balance.
Vitamin C performs most effectively in a relatively low pH environment. However, lower pH levels can increase the likelihood of skin irritation, particularly in products positioned for sensitive skin.
Balancing performance and tolerance becomes a formulation decision rather than a marketing one.
Finally, there is the question of claim language.
While claims such as brightening may be realistic, stronger statements like repairing pigmentation or correcting sun damage may require clinical substantiation or cross into regulatory territory depending on the market.
This is where the feasibility stage becomes valuable.
The goal is not to eliminate the concept; Vitamin C remains one of the most effective and widely recognised cosmetic actives.
The goal is to understand the technical consequences early, so formulation strategy, packaging choices and claim positioning remain aligned.
Because in cosmetic development, ingredients rarely act in isolation.
A single ingredient decision can reshape the entire product system, from stability and packaging to testing requirements and regulatory positioning.
Why this matters before formulation
Formulation rounds are not designed to determine what a product should be.
They are meant to execute a direction that has already been defined.
When that direction is unclear, development often becomes a process of discovery rather than execution. Laboratories begin exploring possibilities, feedback loops expand and small technical issues start influencing larger decisions.
This is where many projects lose momentum.
That scrutiny is visible at the regulatory level. According to the European Commission’s Safety Gate Annual Report (2024), more than 4,100 alerts were recorded in the system, with cosmetics ranking as the most frequently notified product category.(European Commission Safety Gate Report 2024)
Without a clear feasibility stage, product development can quickly become a cycle of adjustments:
• additional formulation rounds
• conflicting feedback on texture or performance
• stability issues discovered late in testing
• packaging changes triggered by formulation constraints
• marketing claims that require revision once the formula reality becomes clearer
None of these issues necessarily mean the product concept is flawed.
More often, they reflect decisions that were never fully examined at the beginning of the process.
A feasibility review reduces this uncertainty by identifying the key constraints, risks and trade-offs before the laboratory begins its work.
One way to understand the role of feasibility reviews is to look at where development problems typically appear in a project timeline.
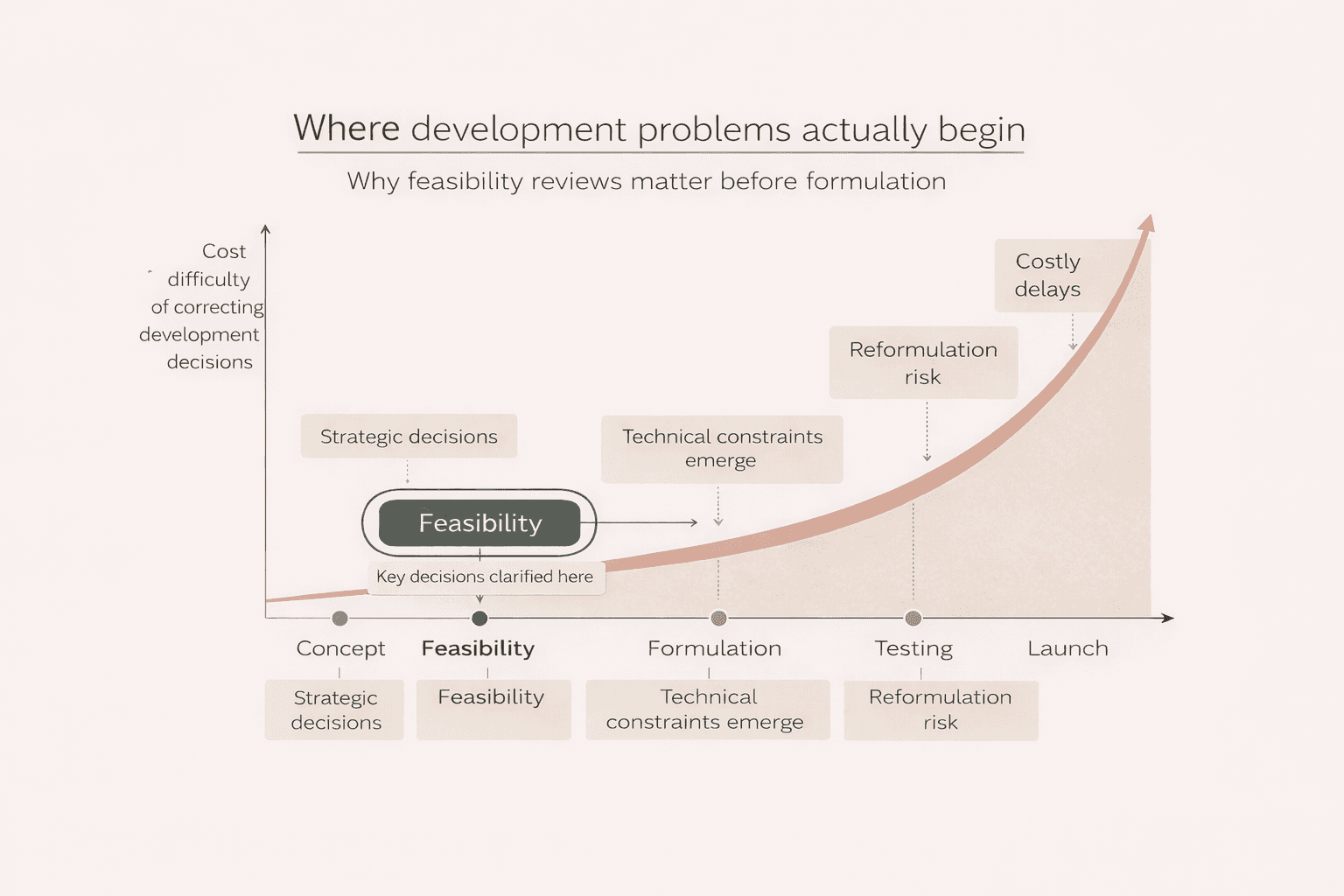
The later a problem is discovered, the more expensive and disruptive it becomes to correct.
Instead of discovering technical limitations during formulation, founders gain a structured understanding of the product’s direction from the start.
The difference is not that complexity disappears. It is that it is identified early when decisions are still flexible and development can move forward with greater clarity.
What Founders Walk Away With
A feasibility review does not replace formulation.
What it provides is clarity before formulation begins.
Founders typically leave the process with a structured understanding of the project:
• whether the concept is technically realistic
• which formulation environment is most suitable
• what ingredient direction is likely to succeed
• how ambitious claims should be framed
• which technical risks may influence development timelines
In other words, the product idea becomes a development direction rather than a hypothesis.
For laboratories, this clarity translates into a more focused formulation process.
For founders, it reduces uncertainty and helps ensure that the decisions shaping the product are made deliberately rather than discovered later.
The Coseer Perspective
At Coseer, a feasibility review is a structured pre-development assessment.
It is designed to clarify whether a product concept is coherent before a laboratory is briefed; across ingredient direction, formulation environment, claim positioning, regulatory considerations and overall development complexity.
We offer Feasibility Reviews in three levels (A-C) depending on the stage and depth of the project, from an initial direction check to a more developed lab-ready brief.
The purpose is clear: to ensure that by the time formulation begins, the product direction has already been examined, challenged and structured.
Because stronger development does not begin with more samples.
It begins with better decisions.

